CO2 Reduction WC
Sippakorn Wannakao's Research
Project Description
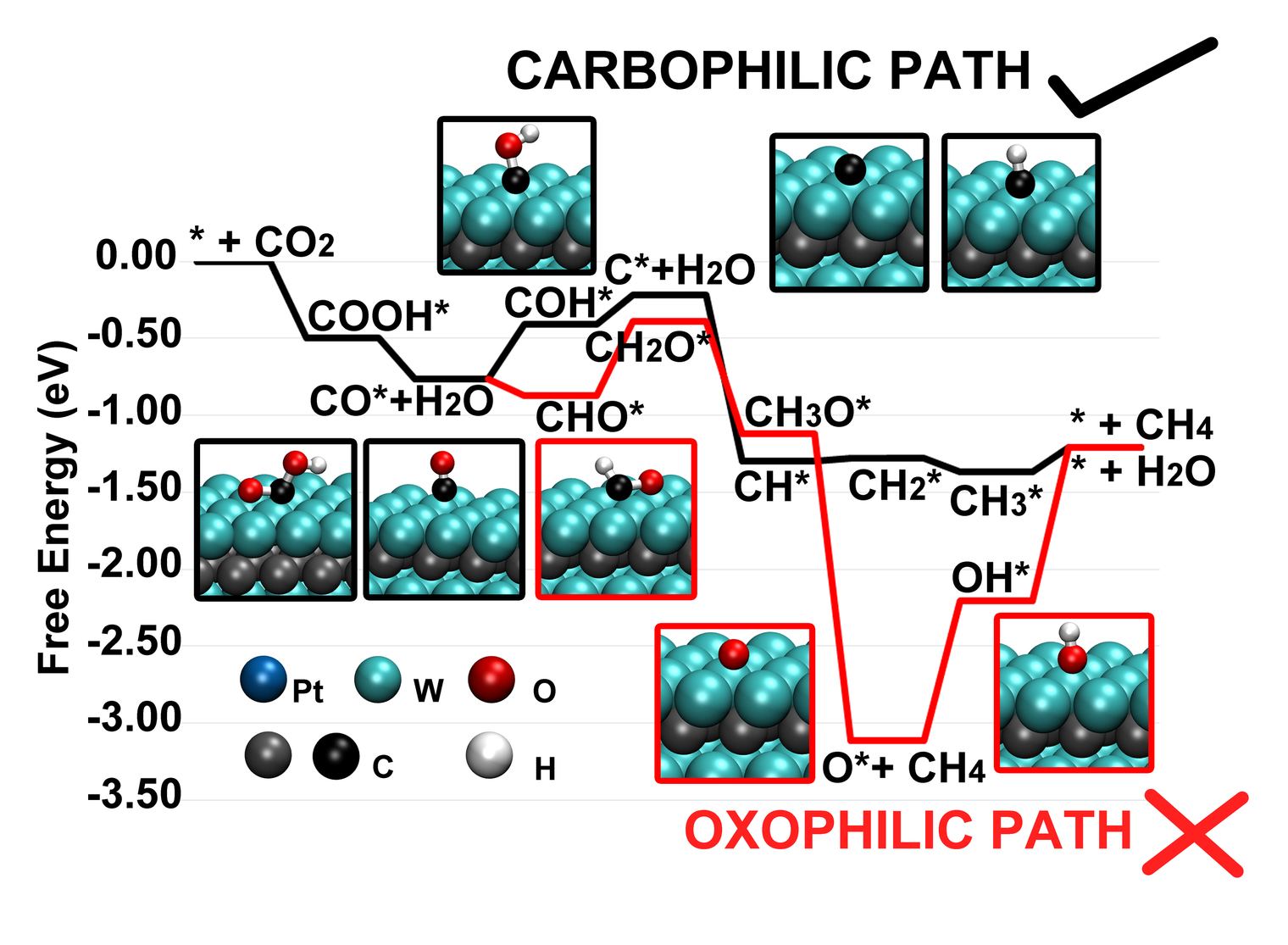
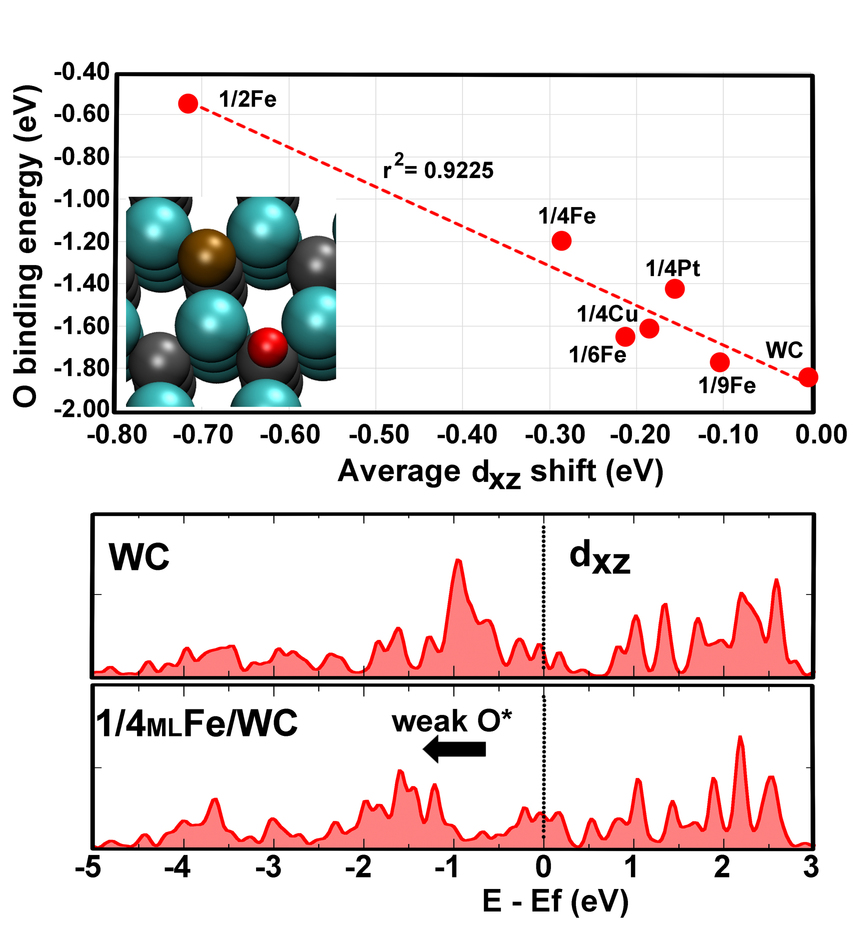
Capture and conversion of carbon dioxide (CO2) into valuable chemicals is a critical challenge in the development of environmental friendly, economic, and renewable energy technologies. Beside precious metals, transition metal carbides (TMCs) have shown promise as effective catalysts, potentially enabling a low-cost alternative platform for concomitant optimization of activity and selectivity. Using first-principles computations, we demonstrate that the behavior of these catalysts is well-described by the weighted positions of individual d-band components, and we postulate that this model can be applied to predict site preference and binding energy trends for reaction intermediates on complex catalyst surfaces with highly non-degenerate d-band components. We show that directional bonding arising from the inherently mixed covalent/metallic character of TMCs plays a key role in governing surface chemistry in bare and coated TMCs. This new understanding can be exploited to tailor catalysts with desirable product selectivity and optimized activation potential by tuning the oxophilicity and carbophilicity of the surface.